China Cosmetic Product Registration/Notification
Overview
The new overarching rule on cosmetics in China – Cosmetic Supervision and Administration Regulation (CSAR) was released by China State Council on June 29, 2020. With CSAR that came into effect on January 1, 2021, the cosmetics industry in China is entering a new era.
Cosmetic products now must be notified/registered with NMPA before being imported and sold legally in China.
Definition of Cosmetic Products
| Aspects | Cosmetics | Not Cosmetics |
| Usage | Spreading, spraying, or other similar ways like rubbing | Injection, digestion, or oral administration |
| Applied body parts | External parts of the human body, such as skin, hair, nails, lips | Oral mucosa or internal genitalia |
| Functions and purpose of use | For cleansing, protecting, beautifying, or improving the appearance | Disease prevention or treatment |
| N.B.:
• Toothpaste is not defined as cosmetics but shall be regulated with reference to general cosmetics regulations. • CSAR does not apply to soaps, excluding those claimed to have special cosmetics efficacy (e.g., whitening). |
||
Classification of Cosmetic Products
Special Cosmetics vs. General Cosmetics
| Special Cosmetic Products | General Cosmetic Products |
| i. Hair dyes
ii. Hair perming products iii. Freckle-removing (whitening) products iv. Anti-hair loss products v. Sunscreens vi. Cosmetics with new efficacy |
Other cosmetics except for special cosmetics. |
| N.B.:
• The five previously classified as special-use cosmetics – hair growth, depilating, breast beauty, slimming, and deodorant products registered before January 1, 2021, can continue to be manufactured, imported, and sold in China during the 5-year transition period. |
|
Domestic Cosmetics vs. Imported Cosmetics
| Domestic cosmetics | The last process of contacting cosmetics contents is completed in Chinese Mainland. |
| Imported cosmetics | The last process of contacting cosmetics contents is completed out of Chinese Mainland. |
| N.B.:
• If the product’s last process of contacting cosmetic contents is completed in Hong Kong S.A.R, Macao S.A.R, or Chinese Taipei, it shall be managed with reference to the requirements of imported cosmetics. |
|
Notification/Registration of Cosmetic Products
| Classification | Special cosmetics | General cosmetics |
| Pre-Market Obligation | Registration | Notification |
| Involved Entities | Registrant (domestic)
Responsible Person (imported) |
Notifier (domestic)
Responsible Person (imported) |
| Competent Authority | NMPA | NMPA or local provincial MPA |
| Certificate | Registration license
(Valid for five years) |
E-certificate
(Permanent) |
| Post-Market Obligation | Registrational renewal | Annual report submission |
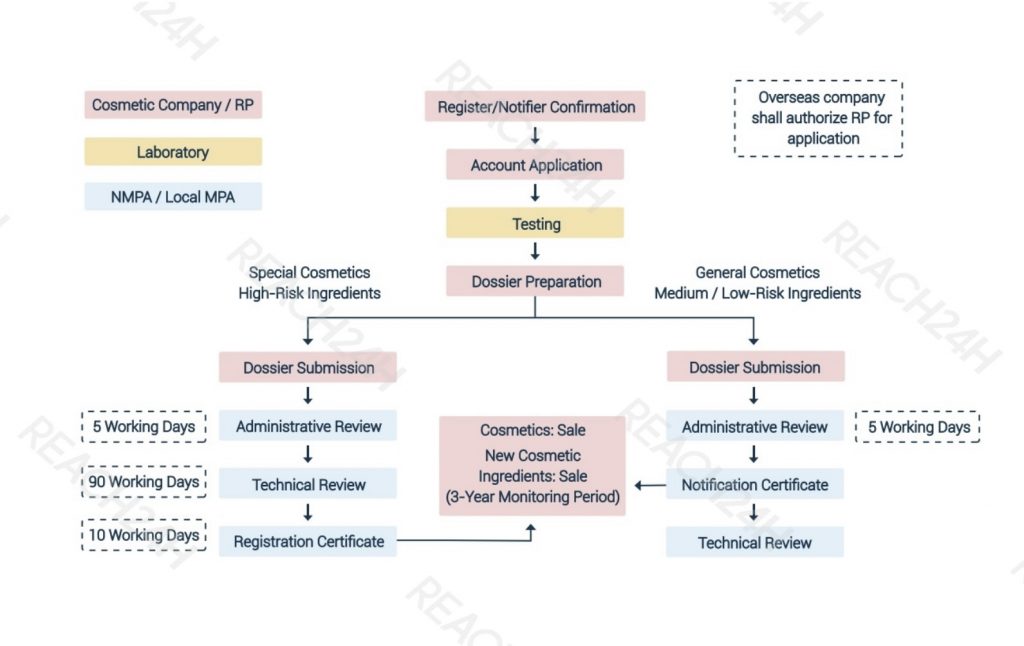
Steps for Imported Cosmetic Registration/Notification
Competent Authorities in China
-
National Medical Product Administration (NMPA)
It is responsible for the registration of imported/domestic special cosmetics, the notification of imported general cosmetics, and the registration and notification of new cosmetic ingredients.
-
State Administration for Market Regulation (SAMR)
Its main responsibilities include market management and quality and safety supervision.
-
General Administration of Customs (GAC)
It is in charge of the inspection of imported and exported goods and quarantine of cosmetics.
Notification/Registration Dossiers
Account Application and User Permission Opening
- Registrant and notifier information form and resume of the person in charge of quality and safety
- Quality management system overview of registrants and notifiers
- Adverse reaction monitoring and evaluation system overview form of registrants and notifiers
- Domestic responsible person information form (applicable to overseas registrants and notifiers)
- The original authorization letter of the domestic responsible person and its original notarial certificate
Registration and Notification Application
- Cosmetics registration and notification information form and related documents (classification code)
- Product name information
- Product formula (ingredient submission code)
- Product executive standards
- Product label sample manuscript
- Product testing report
- Product safety assessment documents
- Other supporting documents
N.B:
- Imported cosmetics are required to submit certifications relating to manufacturing quality control of the overseas manufacturers.
- For products specially produced for the China market without the supporting documents, the applicant shall submit relevant research and test data for Chinese consumers.
Our Services
- Cosmetics Regulatory and Technical Advisory
- Cosmetics Registration/Notification
- China Responsible Person
- Cosmetic Product Safety Report (CPSR)
- Efficacy Claim Evaluation Report
- Labeling and Ingredient Reviews
Why Choose Us?
- Owns a leading online information platform that provides timely insights into regulatory and market intelligence
- Industry-leading expertise in cosmetics compliance consulting services
- Served over 1000+ cosmetics companies worldwide
- Established cooperative relationships with governmental authorities, industry associations, and laboratories
- A professional, global, multilingual, and multicultural team with the capability to deliver services with high efficiency through a systematic approach