QSAR
Quantitative Structure–Activity Relationship (QSAR) models are regression or classification models used in the toxicological sciences.

Generally, the establishment of (Q)SAR model consists of the following five steps:
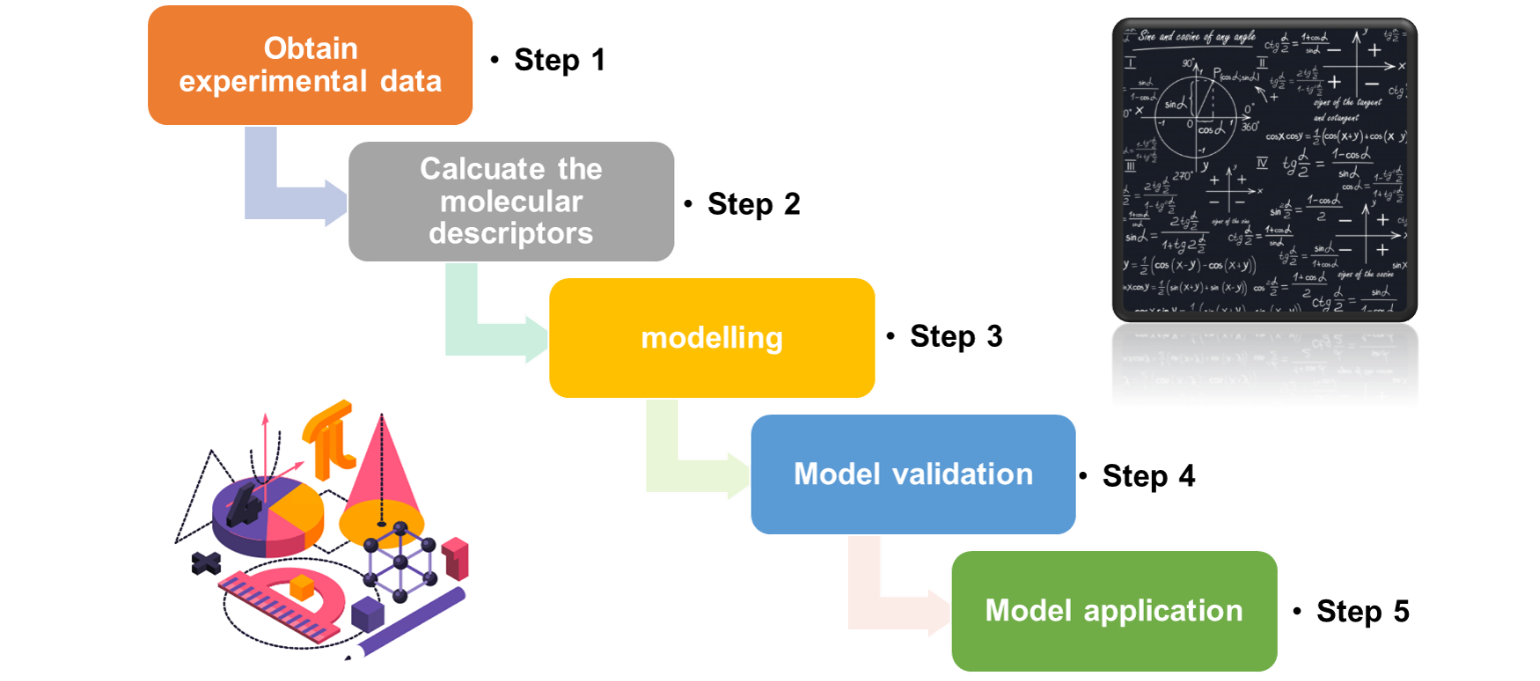
QSAR approaches are usually applied to assess health toxicity or ecotoxicity of new/increased level of impurities existed in a new source of technical materials. The predicted toxicities, especially for genotoxicity, obtained by using QSAR are important data for assessing whether the new active ingredient is toxicological equivalent when compared to its reference source, and the data are required by an increasing number of countries, regions or organizations including the EU, Australia, Brazil, Mexico, Russia, Argentina, etc.

REACH24H offers the best in-silico tools and scientific expertise, which when combined, helps overall assessment of (eco)toxicity, environmental fate, physico-chemical property of impurities or target compounds.
- Publicly available tools: OECD QSAR Toolbox, Toxtree, T.E.S.T, VEGA hub, OPERA, ECOSAR, EPI SuiteTM, etc.
- Commercial tools: Derek NexusTM, Sarah NexusTM, Case Ultra, etc.
- Cheminformatic tools: ChemDraw, RDkit, ChemMine, AMBIT, CPDB/LCDB, etc.


Our Services
QSAR and Read-Across for new/increased level of impurities existed in a new source of technical materials or target compounds.
- Rule-based and Statistical-based QSAR models
- Application of Threshold of Toxicological Concern (TTC)
- Derivation of Health-Based Exposure Limits (HBELs): PDE/ADE, AI, TTC
- Read-Across and chemical similarity searching
- Defined Approaches on skin sensitization (OECD TG 497)
- Virtual screening for lead discovery of pesticide
- QSAR Prediction Report Format (QPRF)
- QSAR Model Report Format (QMRF)
Available Endpoints
- Physico-chemical properties: Log Kow, pKa, BCF, etc.
- Mammalian toxicology: genotoxicity/mutagenicity, carcinogenicity, developmental/reproductive toxicity, acute oral toxicity (rat LD50), endocrine disruption, skin sensitization, etc.
- Ecotoxicology: aquatic toxicity, toxic to honeybees, etc.
- Environmental behavior: persistence, hydrolysis rate, Koc, etc.
- Metabolites and degradation or metabolic pathways of molecules
Genotoxicity and Endocrine Disruption Assessment
- ICH M7 Assessment and control of DNA reactive (mutagenic) impurities.
- in silico evaluation of clastogenicity
- Molecular docking
- Postulate MoA(s) based on adverse outcome pathway (AOP) framework
- Weight of Evidence (WoE)
QSAR model building
- Data cleaning
- Molecular descriptors
- Model selection: linear models & machine learning
- Validation
- Application domain
- Release
Our Strength
- Experienced Expert Team Support
- Strict Quality Control System
- Close Collaboration with Plenty of Experts and Predominant Laboratories
- Good Communication with Competent Authorities
- Fast Response to Clients Inquiries and Controlling Cost
- Obeying the Code of Ethics, Respecting the Confidentiality and Privacy of our Clients
- Excellent Project Management


